 When an atom is formed, several forces are present. The mass defect will be equal to the difference between the calculated mass and the actual mass, as expressed in the following formula: Applied to the helium atom, we get:Īs we can see, mass is lost when the nucleus is formed and stored in the form of energy, which is known as binding energy. What is the mass defect of the helium? To calculate this, we add the masses of the protons and neutrons together. The proton has a mass of 1.00727 amu, while the neutron has a mass of 1.00866 amu. Using this unit, we can say that the helium atom has a mass of 4.0026 amu. The mass of an atom can be expressed by a unit called atomic mass unit (amu), which is the mass of 1/12th of the mass of a neutral, non-bonded carbon. The nuclear mass is just the mass of the atom nucleus.Ī stable helium atom has two protons and two neutrons. A is the mass number, while Z is the atomic number. The total mass of the protons will be the sum of the mass of each proton, mp from n=1 to Z will be the sum of the mass of all neutrons from n=1 to A-Z. The total mass of all neutrons will be the sum of the mass of each neutron mn from n=1 to A-Z. The total mass of the nucleus will be the sum of all neutrons and protons. Here, mn and mp are the individual masses of the protons and neutrons that compose a particular atom. The difference between the total mass of the individual elements in the atom and the atom’s real mass is known as the mass defect. The lost mass is stored in the nucleus as energy. 
If you take the masses of each individual particle that forms the nucleus of an element, its combined weight is larger than the mass of the element’s nucleus. Elements that have more particles in the nucleus are, therefore, more massive and have more charge. Each particle has its own properties, such as charge and mass. These are electrons, neutrons, and protons. Atoms are composed of particles that occur in their nucleus. Mass defect and binding energyĪn example of the energy-mass relationship is found in nuclear physics. However, the mass-energy equivalence can be used with respect to all forms of energy. The mass of a body is a measure of its energy content.Įinstein’s theory only speculated that energy emission in the form of light reduced the amount of the object’s mass. The energy emitted is independent of the body’s characteristics. His results include two important points: Einstein describes how the emission of energy in the form of light reduces the kinetic energy of the body. The Einstein derivationĪlbert Einstein derived his famous equation in 1905 in an article entitled ‘Does the inertia of a body depend upon its energy content’. Here, m is the mass lost in kg, E is the energy in Joules, and c is the velocity of light in a vacuum, which is equal to 3.00 ⋅ 10 ^ 8 m/s. 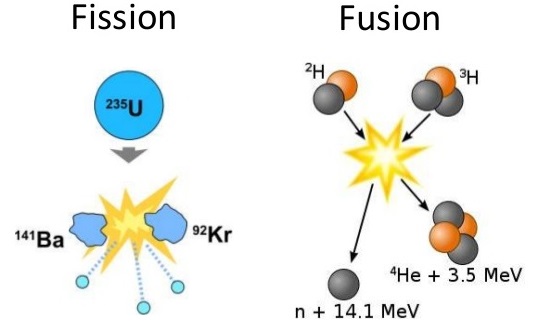
Thompson and Oliver Heaviside observed changes in the mass of an object when it has an electrical charge, a phenomenon that has been described as electromagnetic mass.īut it was Einstein who proposed that when an object emits energy E in the form of electromagnetic radiation, it loses mass equal to the energy divided by the square of the velocity of light. Isaac Newton speculated about matter and light being convertible to each other, and several attempts to relate matter to kinetic energy were made in the following centuries. 
The relation between mass and energy was studied by many scientists before Einstein. Developing technology to harness nuclear fusion as a source of energy for heat and electricity generation is the subject of ongoing research, but whether or not it will be a commercially viable technology is not yet clear because of the difficulty in controlling a fusion reaction.Albert Einstein expressed the concept of energy-mass equivalence Fusion is the source of energy in the sun and stars. Nuclear energy can also be released in nuclear fusion, where atoms are combined or fused together to form a larger atom. This reaction is controlled in nuclear power plant reactors to produce a desired amount of heat. This process is called a nuclear chain reaction. These neutrons continue to collide with other uranium atoms, and the process repeats itself over and over again. More neutrons are also released when a uranium atom splits. During nuclear fission, a neutron collides with a uranium atom and splits it, releasing a large amount of energy in the form of heat and radiation. All nuclear power plants use nuclear fission, and most nuclear power plants use uranium atoms. In nuclear fission, atoms are split apart, which releases energy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
